Vaccines | Free Full-Text | Estimating the Impact of Vaccination Campaigns on Measles Transmission in Somalia
[ad_1]
1. Introduction
In response to these issues, Somalia’s ministries of health partnered with UNICEF and the WHO to conduct measles vaccination campaigns, integrated with other health services like vitamin A supplementation, across the country in 2018, 2019–2020, and in 2022. Together, these campaigns delivered more than 10 million measles vaccines to children under 10. This was an ambitious immunization effort in a uniquely challenging setting, complicated further by the need for COVID-19 safety precautions. Documenting and understanding the campaigns’ implementation details and epidemiological implications represents an opportunity for us to gain insight into vaccine delivery in fragile settings.
To that end, in this Communication, we use modern disease modeling methods to assess the impact of these campaigns on measles transmission in Somalia. Briefly, taking a comprehensive epidemiological dataset as input, including data from case-based surveillance, vaccine registries, and serological surveys, we construct a Somalia-specific model of measles transmission from 2018 to 2023. We use this model to estimate the impact campaign doses had on measles susceptibility, and we compare the current situation to a model-based counter-factual epidemiology without the campaigns. This assessment illustrates that Somalia’s vaccination efforts have prevented thousands of measles infections from 2019 to 2023.
2. Measles Epidemiology and Immunization in Somalia
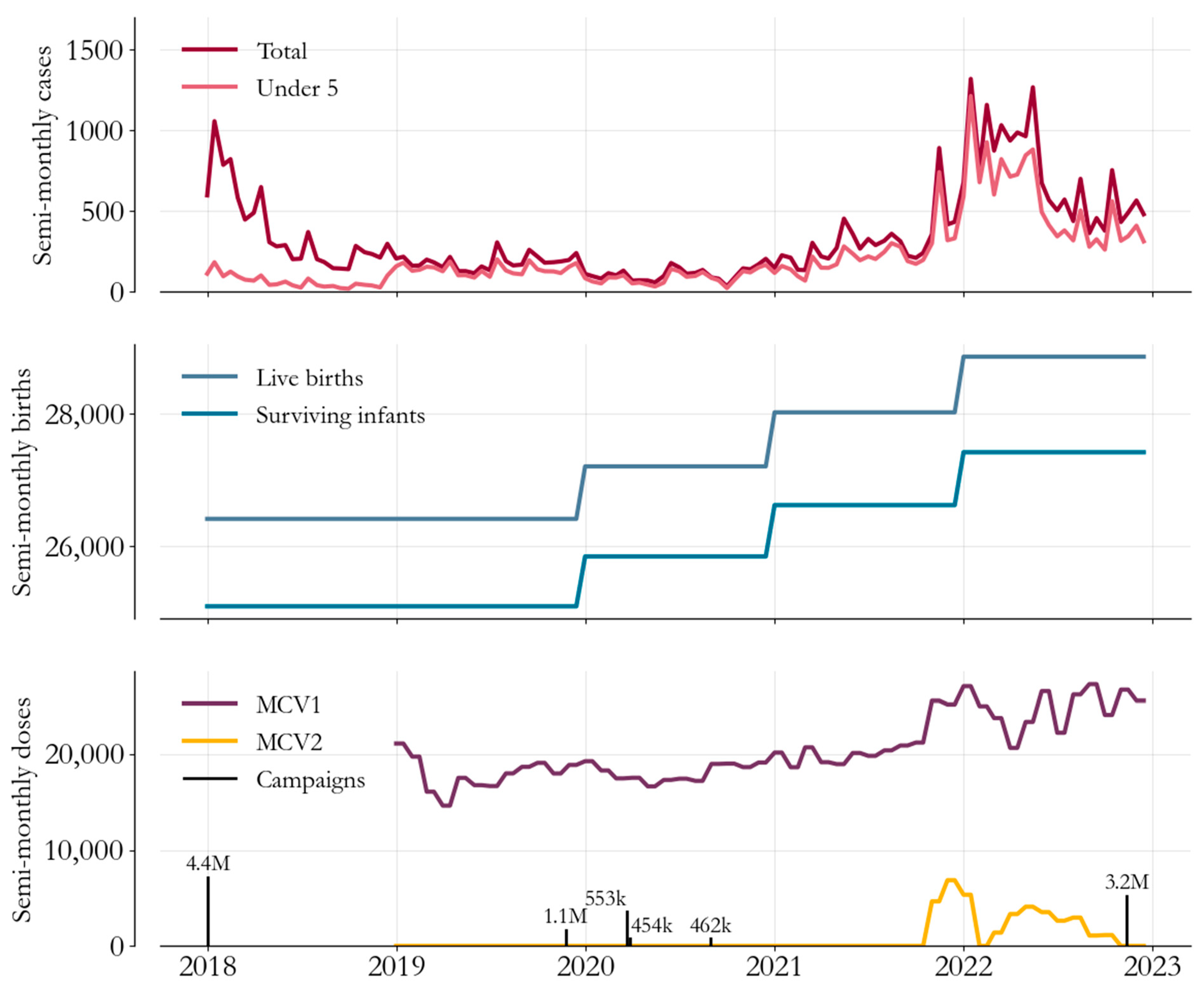
In the second panel, the administrative estimates of yearly live births and surviving infants are plotted on the same semi-monthly increments as cases, under the assumption that the seasonality in birth rates is negligible. We see that birth cohorts have grown in size since 2020, in turn increasing the size of the population at risk of measles infection.
Both the 2018 and 2022 campaigns had a nationwide scope, with the 2018 campaign targeting children up to age 10 and the 2022 campaign up to 5. Meanwhile, in contrast, partly for security reasons, the efforts in 2019–2020, also targeting children under 5, were implemented in geographically distinct phases. All southern states except Banadir implemented the campaign in November 2019; Puntland and Somaliland implemented in the last week of March 2020, and Banadir implemented in August and September 2020.
To disentangle these distinct contributions, we assume that, conditional upon being unvaccinated, the age-at-infection is log-normally distributed. The resulting model, with an immunity component taken from the MCV1 data above, under the assumption that MCV1 seroconverts 85% of the time, is illustrated in beige. The binned fit (red dashed line) captures the serological data and gives an inferred estimate of the age-at-infection distribution.
3. Transmission Modeling
4. From Campaign Immunity to Averted Burden
5. Conclusions
This study makes it clear that global partnerships with national governments and local implementing agencies can deliver vaccines and save lives in the face of staggering complexity. The protracted nature of Somalia’s crises has led to a fragile health system with fragmented immunization. The planning, organization, and implementation of successful campaigns against measles in this country in particular, reaching children living in inaccessible and security-compromised areas, is a show of exceptional resilience, strength, and determination.
Author Contributions
N.T. led the project, formulated the model, analyzed the data, and led the writing. P.L., H.A., M.F., S.M.M.R.M., Q.H., N.B.-Z., P.M., M.S., M.A.J., M.D. and A.H.A.A. contributed to the project’s conceptualization, data analysis, and general technical inputs. P.L., M.F., S.M.M.R.M., S.Y.S., P.O., A.M. and A.S.B. all provided technical input to the writing and distillation of the modeling results. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
The data presented in this study are available on request from the corresponding author (N.T.), given permission from the Federal Ministry of Health, Somalia. The data are not publicly available for privacy reasons.
Conflicts of Interest
The authors declare no conflicts of interest. P.L., H.A., N.B.-Z., M.F., Q.H., S.M.M.R.M., A.M., P.O., A.S.B. and S.Y.S, work for the World Health Organization (WHO). The authors alone are responsible for the views expressed in this publication and they do not necessarily represent the decisions, policy or views of the WHO.
References
- Measles vaccines: WHO position paper—April 2017. Wkly Epidemiol Rec. 2017, 92, 205–228.
- World Health Organization. Immunization Agenda 2030: A Global Strategy to Leave No One Behind. Available online: https://cdn.who.int/media/docs/default-source/immunization/strategy/ia2030/ia2030-draft-4-wha_b8850379-1fce-4847-bfd1-5d2c9d9e32f8.pdf?sfvrsn=5389656e_69&download=true (accessed on 26 January 2023).
- McLean, A.R.; Anderson, R.M. Measles in developing countries Part I. Epidemiological parameters and patterns. Epidemiol. Infect. 1988, 100, 111–133. [Google Scholar] [CrossRef] [PubMed]
- Wolfson, L.J.; Grais, R.F.; Luquero, F.J.; Birmingham, M.E.; Strebel, P.M. Estimates of measles case fatality ratios: A comprehensive review of community-based studies. Int. J. Epidemiol. 2009, 38, 192–205. [Google Scholar] [CrossRef] [PubMed]
- Integrated Food Security Phase Classification. Somalia: Acute Malnutrition Situation July–September 2022 and Projection for October–December 2022. Available online: https://www.ipcinfo.org/ipc-country-analysis/details-map/en/c/1155886/?iso3=SOM (accessed on 26 January 2023).
- Mohamud, A.K.; Ahmed, O.A.; Ali, I.A.; Dirie, N.I. Demographical, clinical, and complication differences between vaccinated and unvaccinated hospitalized children with measles in Mogadishu Somalia: A hospital-based retrospective cohort study. Ann. Med. Surg. 2023, 85, 1550–1555. [Google Scholar] [CrossRef] [PubMed]
- Nchasi, G.; Paul, I.K.; Sospeter, S.B.; Mallya, M.R.; Ruaichi, J.; Malunga, J. Measles outbreak in sub-Saharan Africa amidst COVID-19: A rising concern, efforts, challenges, and future recommendations. Ann. Med. Surg. 2022, 81, 104264. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.populationpyramid.net/somalia/2018/ (accessed on 11 May 2023).
- Carcelen, A.C.; Winter, A.K.; Moss, W.J.; Chilumba, I.; Mutale, I.; Chongwe, G.; Monze, M.; Mulundu, G.; Nkamba, H.; Mwansa, F.D.; et al. Leveraging a national biorepository in Zambia to assess measles and rubella immunity gaps across age and space. Sci. Rep. 2022, 12, 10217. [Google Scholar] [CrossRef] [PubMed]
- Prada, J.M.; Metcalf, C.J.E.; Ferrari, M.J. Improving measles incidence inference using age-structured serological data. Epidemiol. Infect. 2018, 146, 1699–1706. [Google Scholar] [CrossRef]
- Thakkar, N.; Gilani, S.S.A.; Hasan, Q.; McCarthy, K.A. Decreasing measles burden by optimizing campaign timing. Proc. Natl. Acad. Sci. USA 2019, 116, 11069–11073. [Google Scholar] [CrossRef] [PubMed]
- Thakkar, N. A modeling approach for estimating dynamic measles case detection rates. arXiv 2022, arXiv:2202.11222. [Google Scholar]
- Finkenstädt, B.F.; Grenfell, B.T. Time series modelling of childhood diseases: A dynamical systems approach. J. R. Stat. Soc. Ser. C Appl. Stat. 2000, 49, 187–205. [Google Scholar] [CrossRef]
- Ferrari, M.J.; Grais, R.F.; Bharti, N.; Conlan, A.J.K.; Bjørnstad, O.N.; Wolfson, L.J.; Guerin, P.J.; Djibo, A.; Grenfell, B.T. The dynamics of measles in sub-Saharan Africa. Nature 2008, 451, 679–684. [Google Scholar] [CrossRef]
- Portnoy, A.; Jit, M.; Ferrari, M.; Hanson, M.; Brenzel, L.; Verguet, S. Estimates of case-fatality ratios of measles in low-income and middle-income countries: A systematic review and modelling analysis. Lancet Glob. Health 2019, 7, e472–e481. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, M.; Frey, K.; Hagedorn, B.; Oteri, A.; Yahya, A.; Hamisu, M.; Mogekwu, F.; Shuaib, F.; McCarthy, K.A.; Chabot-Couture, G. Optimization of frequency and targeting of measles supplemental immunization activities in Nigeria: A cost-effectiveness analysis. Vaccine 2019, 37, 6039–6047. [Google Scholar] [CrossRef] [PubMed]
- Grais, R.; Conlan, A.; Ferrari, M.; Djibo, A.; Le Menach, A.; Bjørnstad, O.; Grenfell, B.T. Time is of the essence: Exploring a measles outbreak response vaccination in Niamey, Niger. J. R. Soc. Interface 2008, 5, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Carter, A.; Msemburi, W.; Sim, S.Y.; Gaythorpe, K.A.; Lambach, P.; Lindstrand, A.; Hutubessy, R. Modeling the impact of vaccination for the immunization Agenda 2030: Deaths averted due to vaccination against 14 pathogens in 194 countries from 2021 to 2030. Vaccine. Available online: https://www.sciencedirect.com/science/article/pii/S0264410X2300854X (accessed on 1 August 2023).
Figure 1.
Measles cases, births, and vaccinations, Somalia, 2018–2023. Top panel: Case-based surveillance data (red) illustrate the concentration of burden in children under 5 (pink). Middle panel: estimates of birth rates show a recent growth in birth cohort size. Bottom panel: routine first (purple) and second (yellow) dose delivery has been augmented by vaccine campaigns (black bars).
Figure 1.
Measles cases, births, and vaccinations, Somalia, 2018–2023. Top panel: Case-based surveillance data (red) illustrate the concentration of burden in children under 5 (pink). Middle panel: estimates of birth rates show a recent growth in birth cohort size. Bottom panel: routine first (purple) and second (yellow) dose delivery has been augmented by vaccine campaigns (black bars).
Figure 2.
Measles serological survey, Somalia, 2021. Serological data (black) show that 95% of Somalians have immunity against measles before turning 30. This data give us insight into the balance between vaccination- and infection-derived immunity.
Figure 2.
Measles serological survey, Somalia, 2021. Serological data (black) show that 95% of Somalians have immunity against measles before turning 30. This data give us insight into the balance between vaccination- and infection-derived immunity.
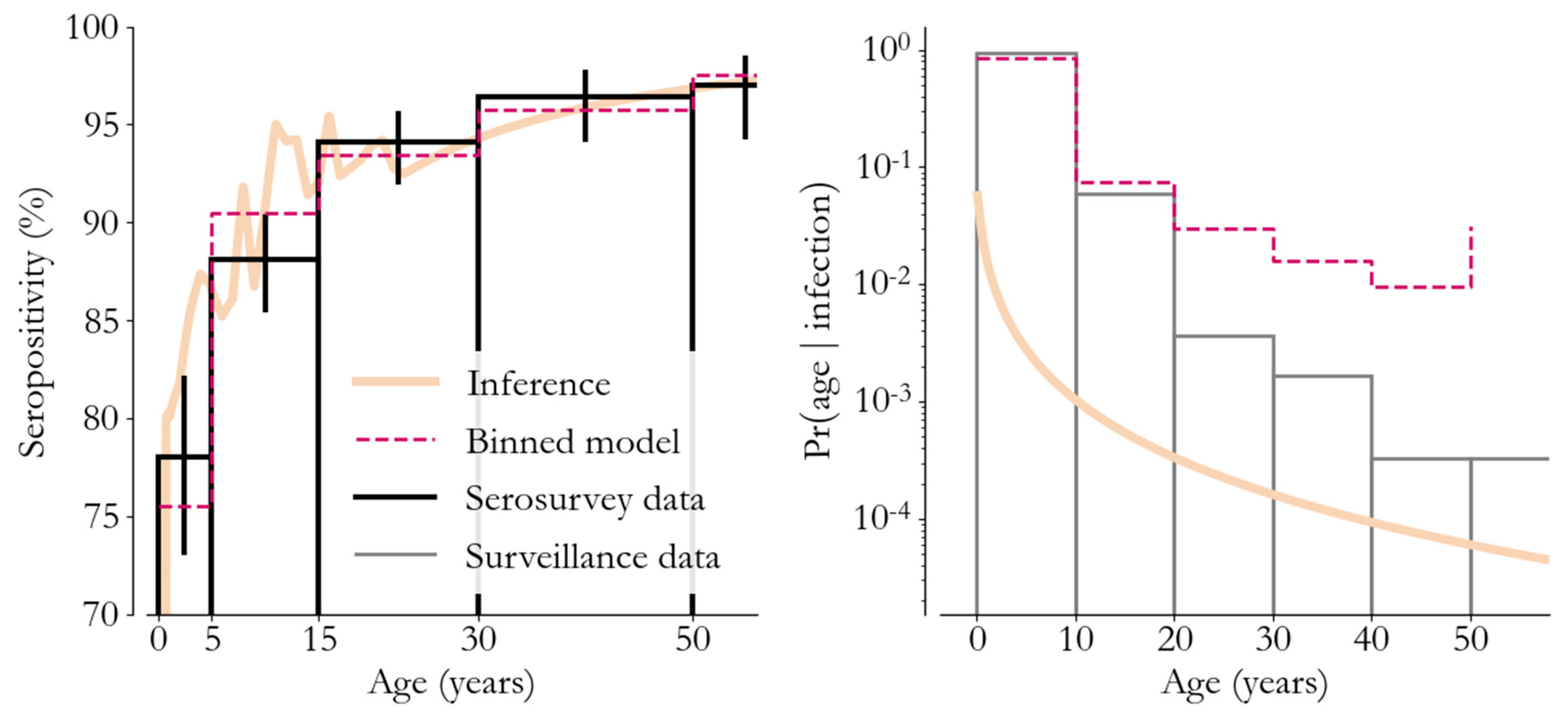
Figure 3.
Modelling measles transmission, Somalia, 2018–2023. Top panel: transmission model samples (mean red line, 95% confidence interval shaded) closely follow observed cases (black dots). Middle panel: the fraction of the population susceptible over time incorporates vaccination campaigns (blue) and agrees with the serological survey results (black line, 95% confidence interval shaded). Bottom panel: the fraction of campaign doses that immunized a susceptible child varies across campaigns (blue shading groups campaign efforts).
Figure 3.
Modelling measles transmission, Somalia, 2018–2023. Top panel: transmission model samples (mean red line, 95% confidence interval shaded) closely follow observed cases (black dots). Middle panel: the fraction of the population susceptible over time incorporates vaccination campaigns (blue) and agrees with the serological survey results (black line, 95% confidence interval shaded). Bottom panel: the fraction of campaign doses that immunized a susceptible child varies across campaigns (blue shading groups campaign efforts).
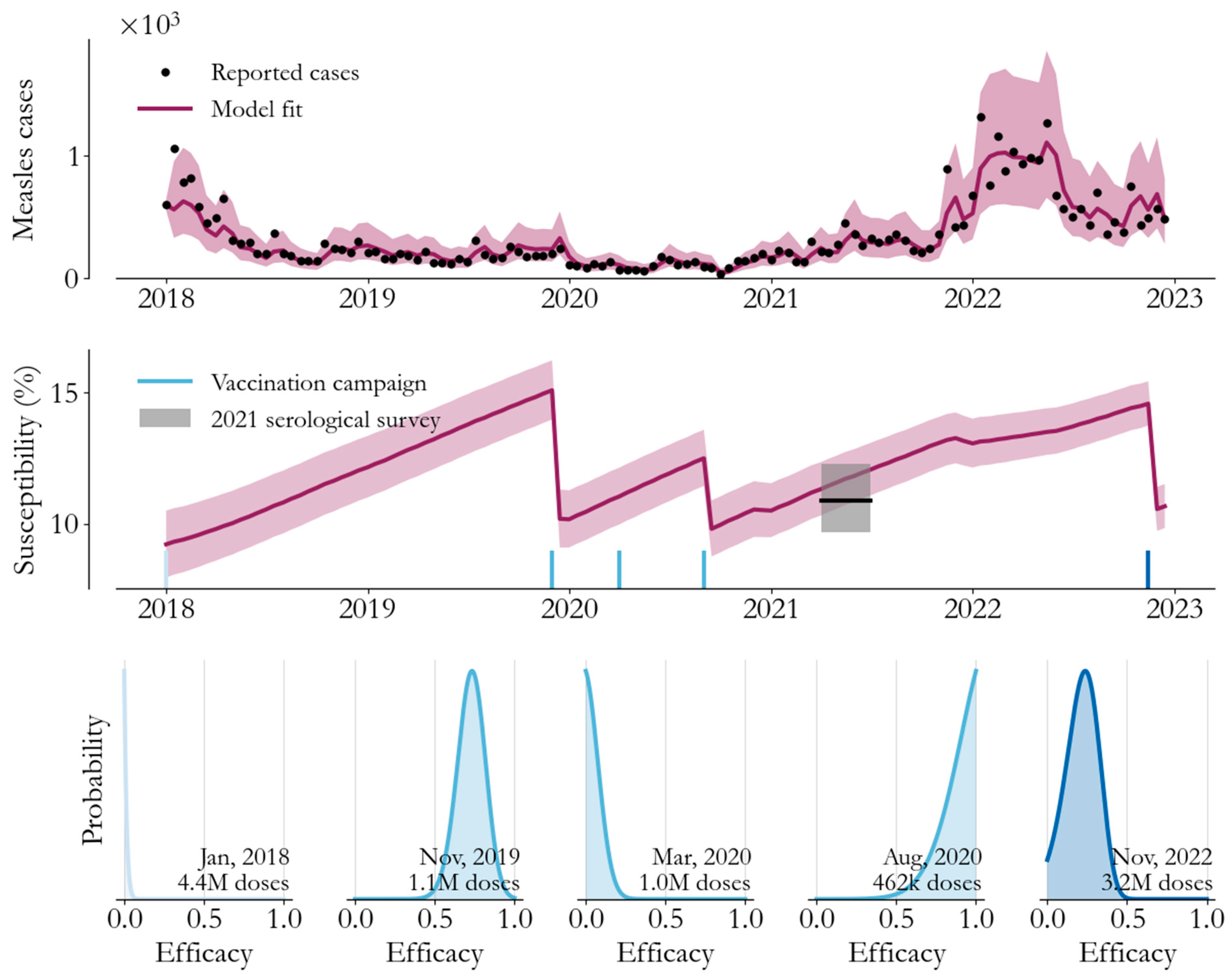
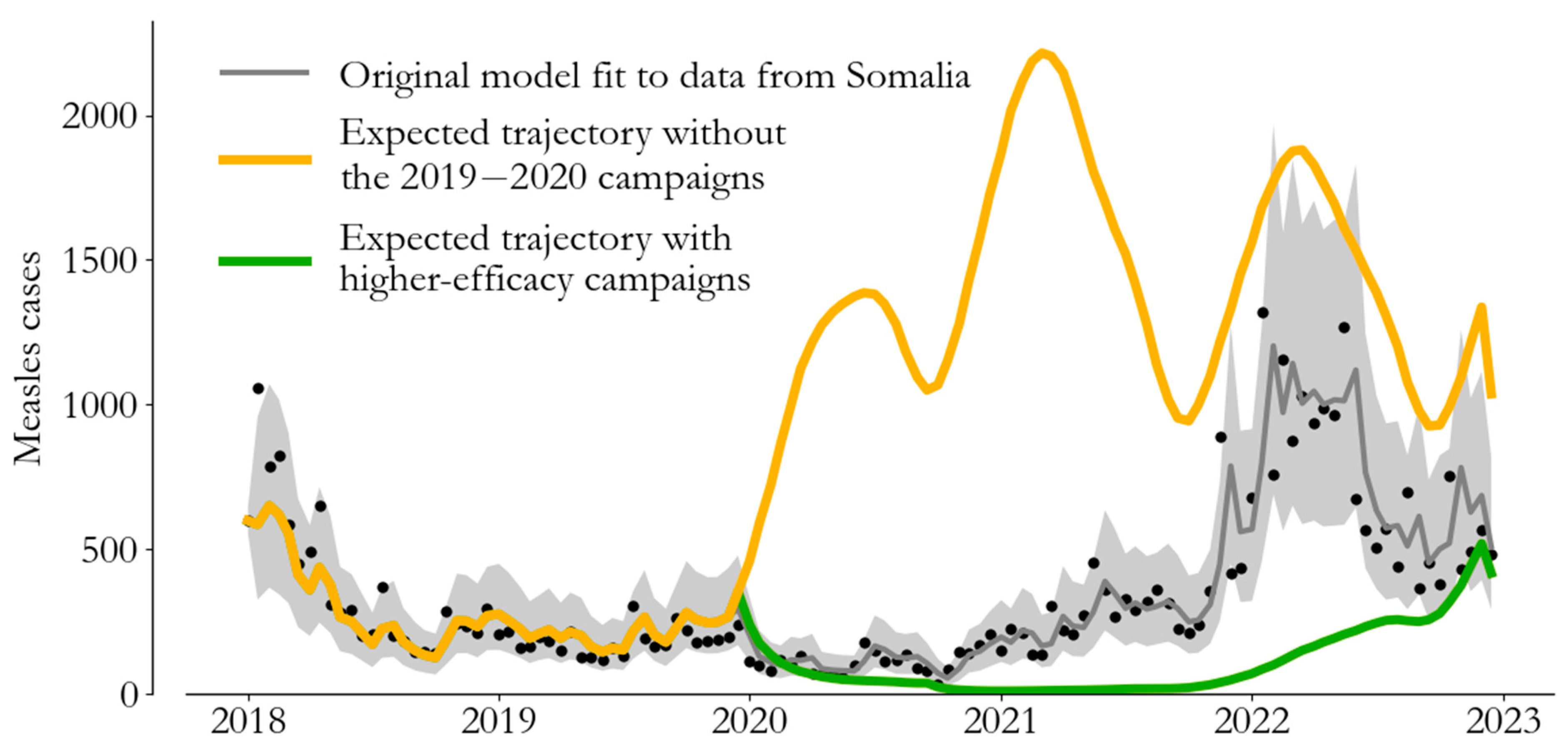
Figure 4.
Model-based counter-factual epidemiology. We can compare the fitted model (grey) to alternative scenarios without the 2019–2020 campaigns (yellow) and with 50% more effective 2019–2020 campaigns (green) to estimate the impact in terms of averted burden.
Figure 4.
Model-based counter-factual epidemiology. We can compare the fitted model (grey) to alternative scenarios without the 2019–2020 campaigns (yellow) and with 50% more effective 2019–2020 campaigns (green) to estimate the impact in terms of averted burden.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
[ad_2]