Healthcare | Free Full-Text | Augmented Articulating Spacers in Infected Total Knee Arthroplasty: Surgical Technique
[ad_1]
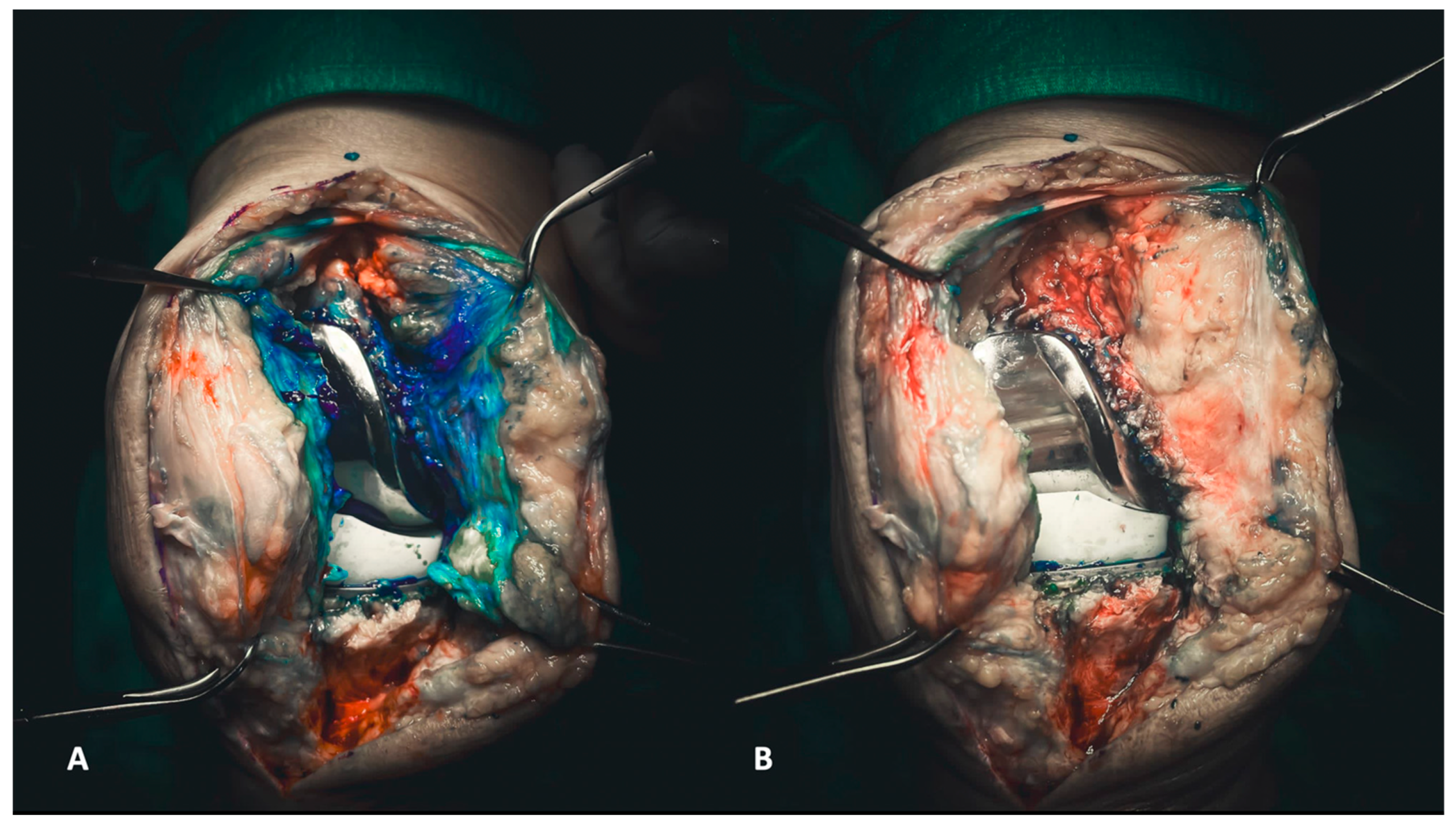
Figure 1.
(A,B) Injecting methylene blue into the joint enables the precise delineation of targeted tissues for subsequent debridement (A). Consequently, the debridement process becomes more discernible, allowing the surgeon to visually assess the entirety of the debridement directly (B).
Figure 1.
(A,B) Injecting methylene blue into the joint enables the precise delineation of targeted tissues for subsequent debridement (A). Consequently, the debridement process becomes more discernible, allowing the surgeon to visually assess the entirety of the debridement directly (B).
Figure 2.
(A,B) The removal of the femoral component must be executed with the goal of preserving the entire bone stock for revision. Each surface of the femoral component needs to be carefully separated, both on the medial and lateral surfaces and the posterior surface (A), to ensure optimal preservation (B).
Figure 2.
(A,B) The removal of the femoral component must be executed with the goal of preserving the entire bone stock for revision. Each surface of the femoral component needs to be carefully separated, both on the medial and lateral surfaces and the posterior surface (A), to ensure optimal preservation (B).
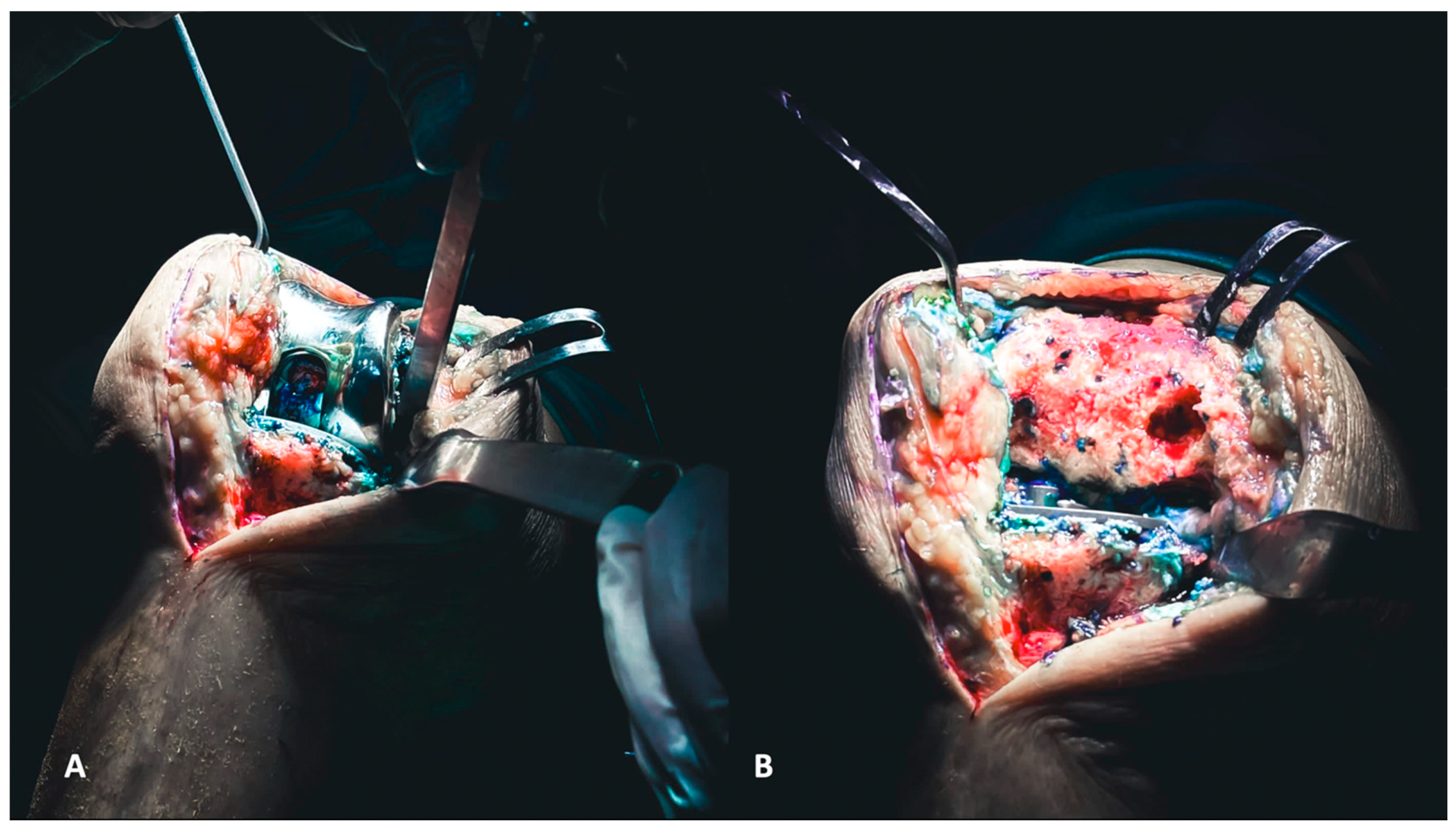
Figure 3.
(A,B) The removal of tibial components, as for the femoral counterparts, requires careful execution to preserve the bone stock. The utilization of a reciprocating saw facilitates the separation of the component from the tibial plateau (A). Additionally, employing a pointed blunt impactor can aid in the removal process by hammering it from below (B).
Figure 3.
(A,B) The removal of tibial components, as for the femoral counterparts, requires careful execution to preserve the bone stock. The utilization of a reciprocating saw facilitates the separation of the component from the tibial plateau (A). Additionally, employing a pointed blunt impactor can aid in the removal process by hammering it from below (B).
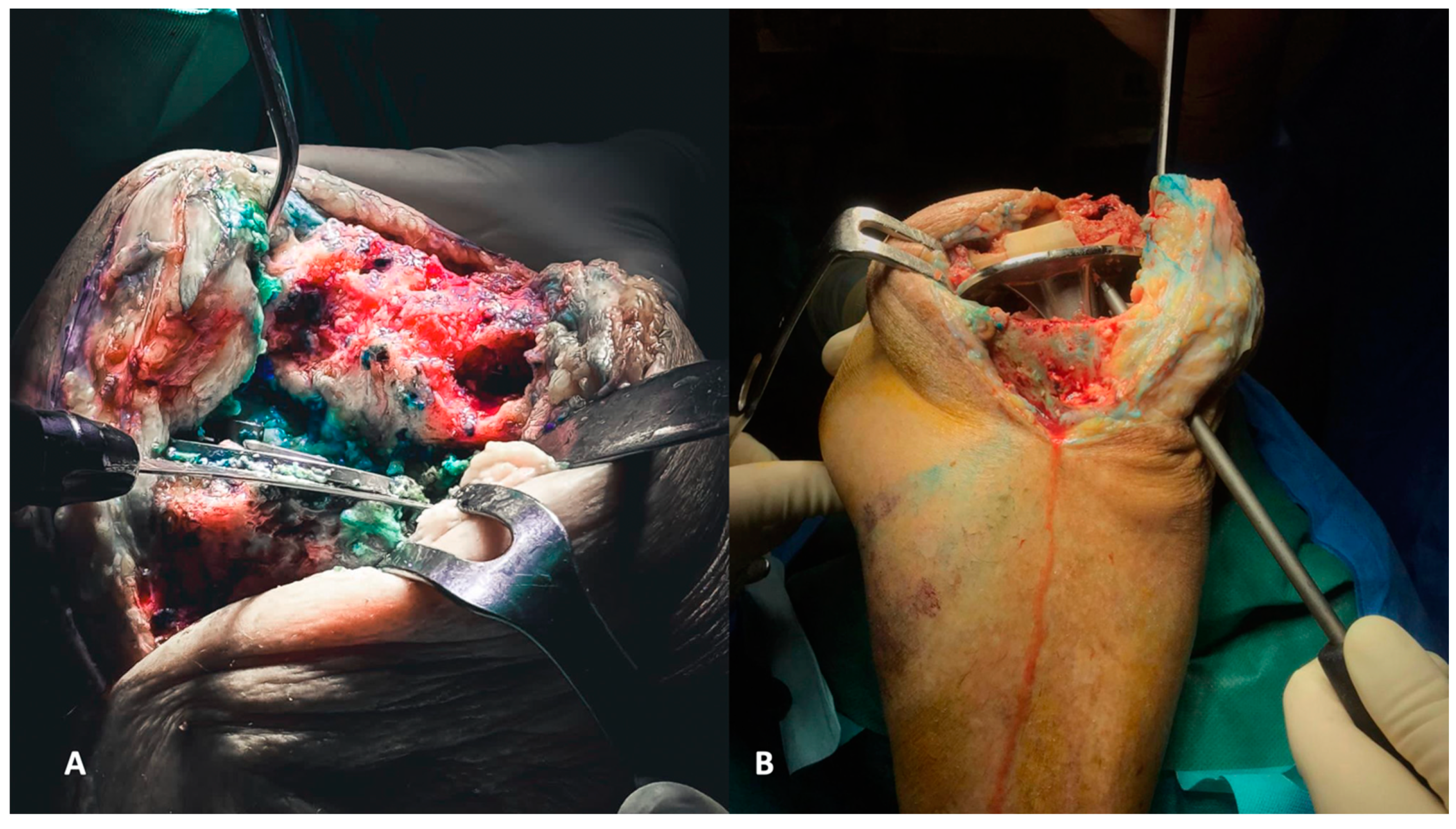
Figure 4.
(A,B) Evaluation of the femoral (A) and tibial (B) axis through extra medullar methods.
Figure 4.
(A,B) Evaluation of the femoral (A) and tibial (B) axis through extra medullar methods.
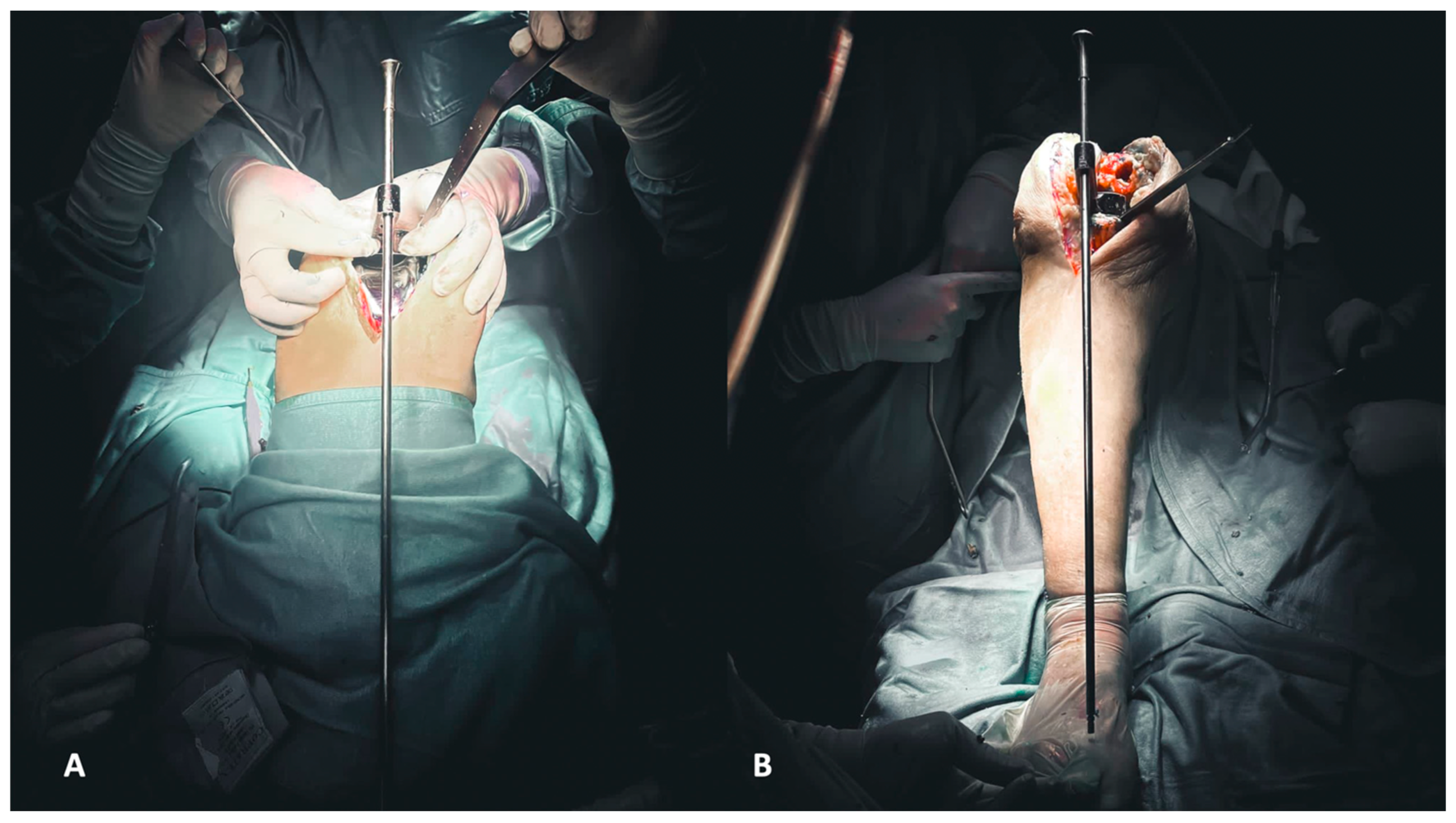
Figure 5.
(A,B) Evaluation of flexion (A) and extension (B) gaps.
Figure 5.
(A,B) Evaluation of flexion (A) and extension (B) gaps.
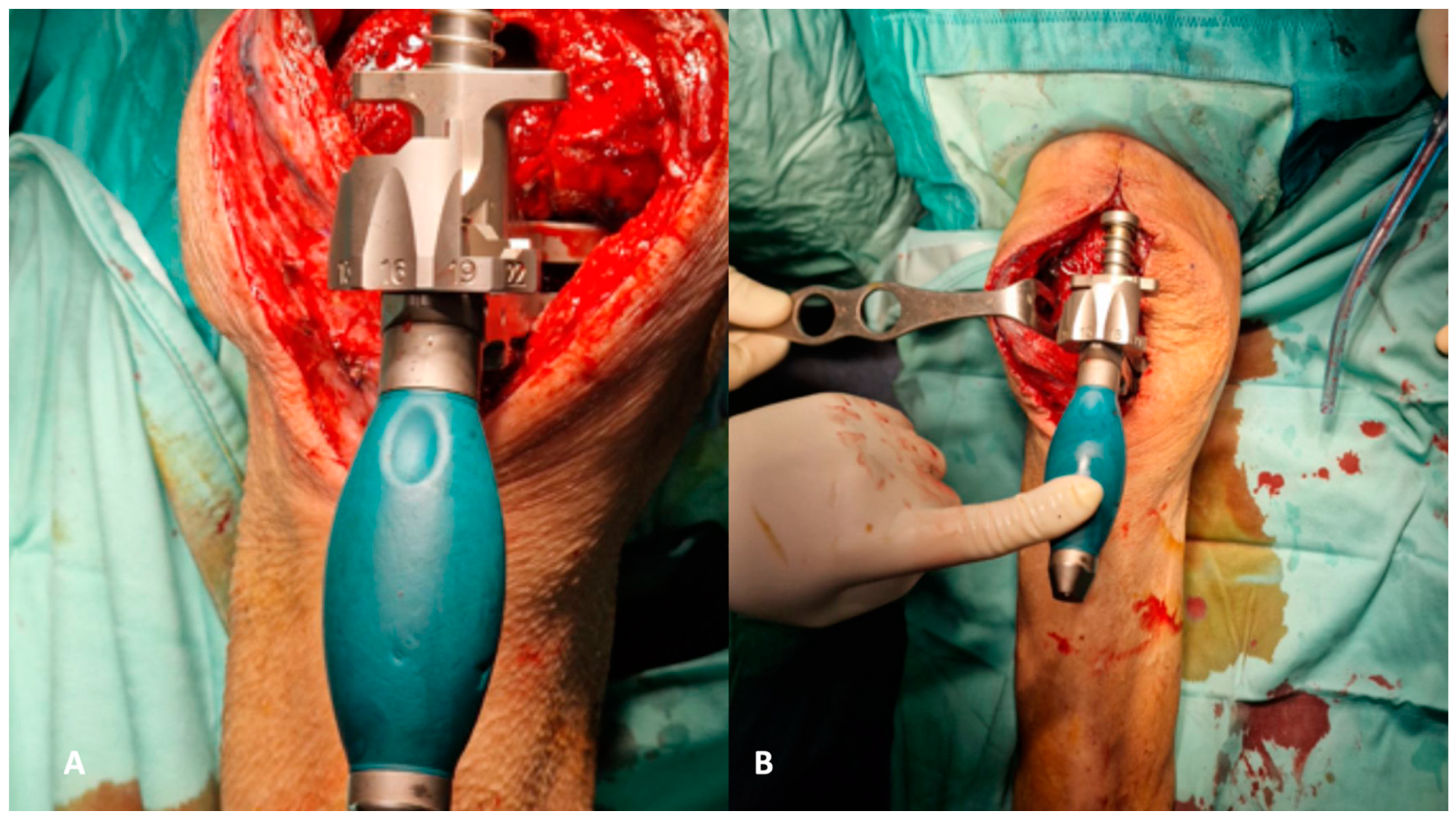
Figure 6.
(A–E) Angular deformities can be addressed using hand-made antibiotic-loaded cement asymmetrical wedges applied to the medial or lateral tibial platform (A). When the tibial platform should be proximalized to fill the gap, a symmetric cemented augment should be prepared (B). To address metaphyseal bone defects, it is important to prepare and use a cemented filler shaped like a cone (C). Like the tibia, angular deformities on the femur’s coronal and traversal planes should be addressed using cemented augments to apply on distal or posterior femoral condyles (D) and self-made stem (rods, wires, or screws) to address the femoral and tibial canal (E).
Figure 6.
(A–E) Angular deformities can be addressed using hand-made antibiotic-loaded cement asymmetrical wedges applied to the medial or lateral tibial platform (A). When the tibial platform should be proximalized to fill the gap, a symmetric cemented augment should be prepared (B). To address metaphyseal bone defects, it is important to prepare and use a cemented filler shaped like a cone (C). Like the tibia, angular deformities on the femur’s coronal and traversal planes should be addressed using cemented augments to apply on distal or posterior femoral condyles (D) and self-made stem (rods, wires, or screws) to address the femoral and tibial canal (E).

Figure 7.
(A,B) Establishing the foundation involves addressing the tibial plateau. Initially, positioning the cone addresses metaphyseal cavitary defects (A), followed by the placement of symmetrical and asymmetrical augments both above (B).
Figure 7.
(A,B) Establishing the foundation involves addressing the tibial plateau. Initially, positioning the cone addresses metaphyseal cavitary defects (A), followed by the placement of symmetrical and asymmetrical augments both above (B).
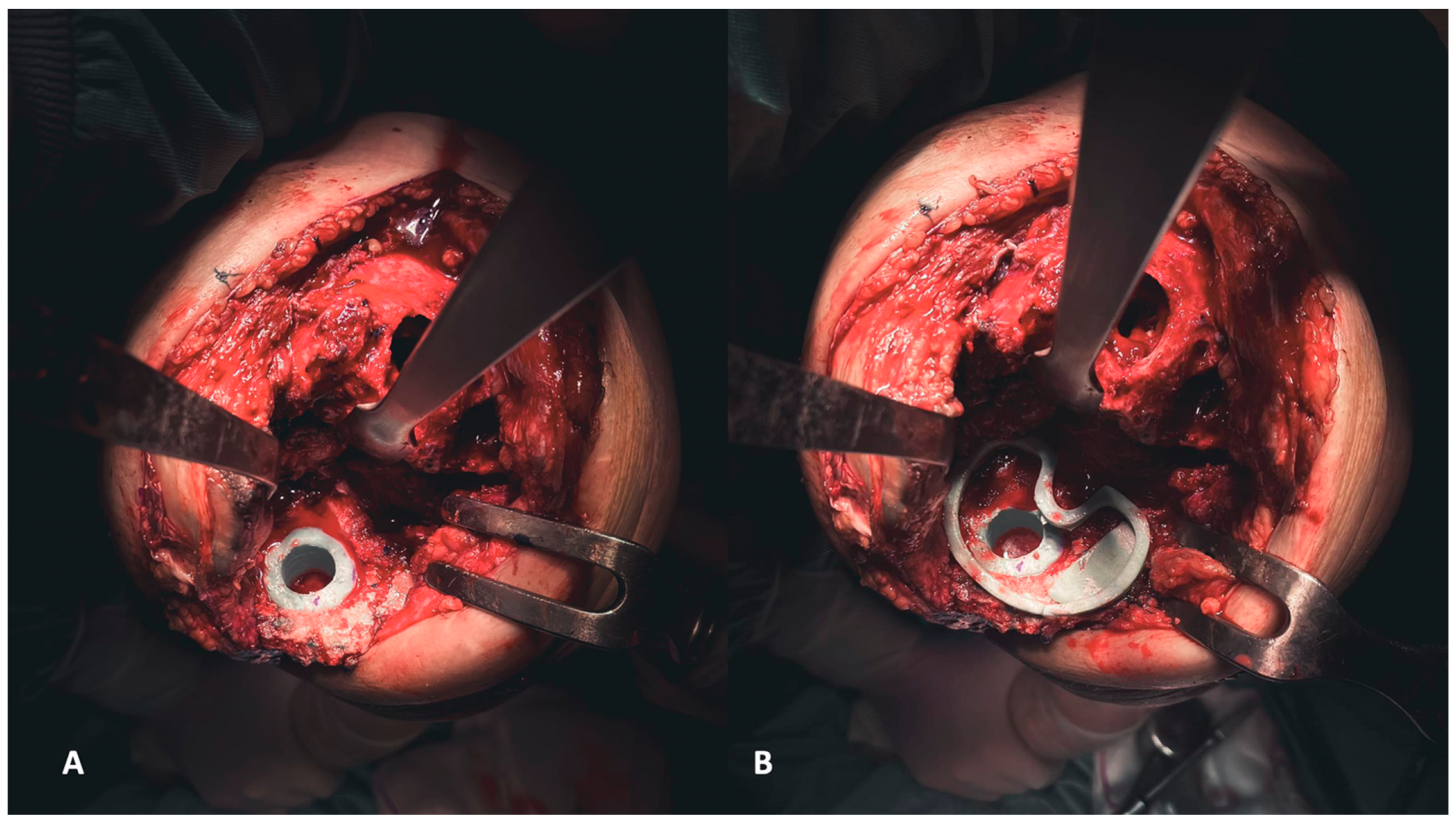
Figure 8.
(A,B) The definitive components for the femur, accompanied by corresponding augments (A), and for the tibia. The latter involves a self-made stem created by inserting a screw into the polyethylene liner, subsequently covered in cement (B).
Figure 8.
(A,B) The definitive components for the femur, accompanied by corresponding augments (A), and for the tibia. The latter involves a self-made stem created by inserting a screw into the polyethylene liner, subsequently covered in cement (B).
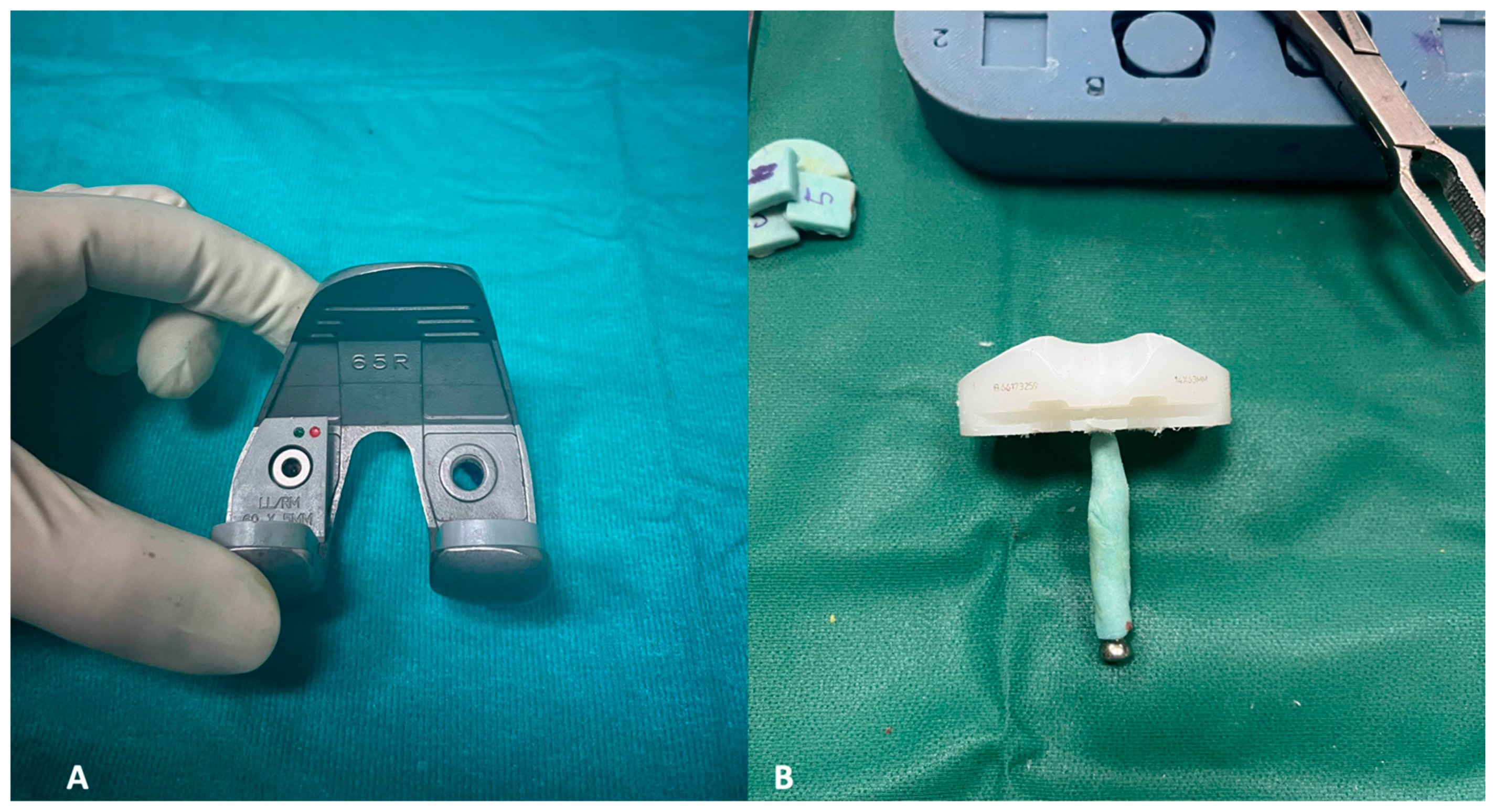
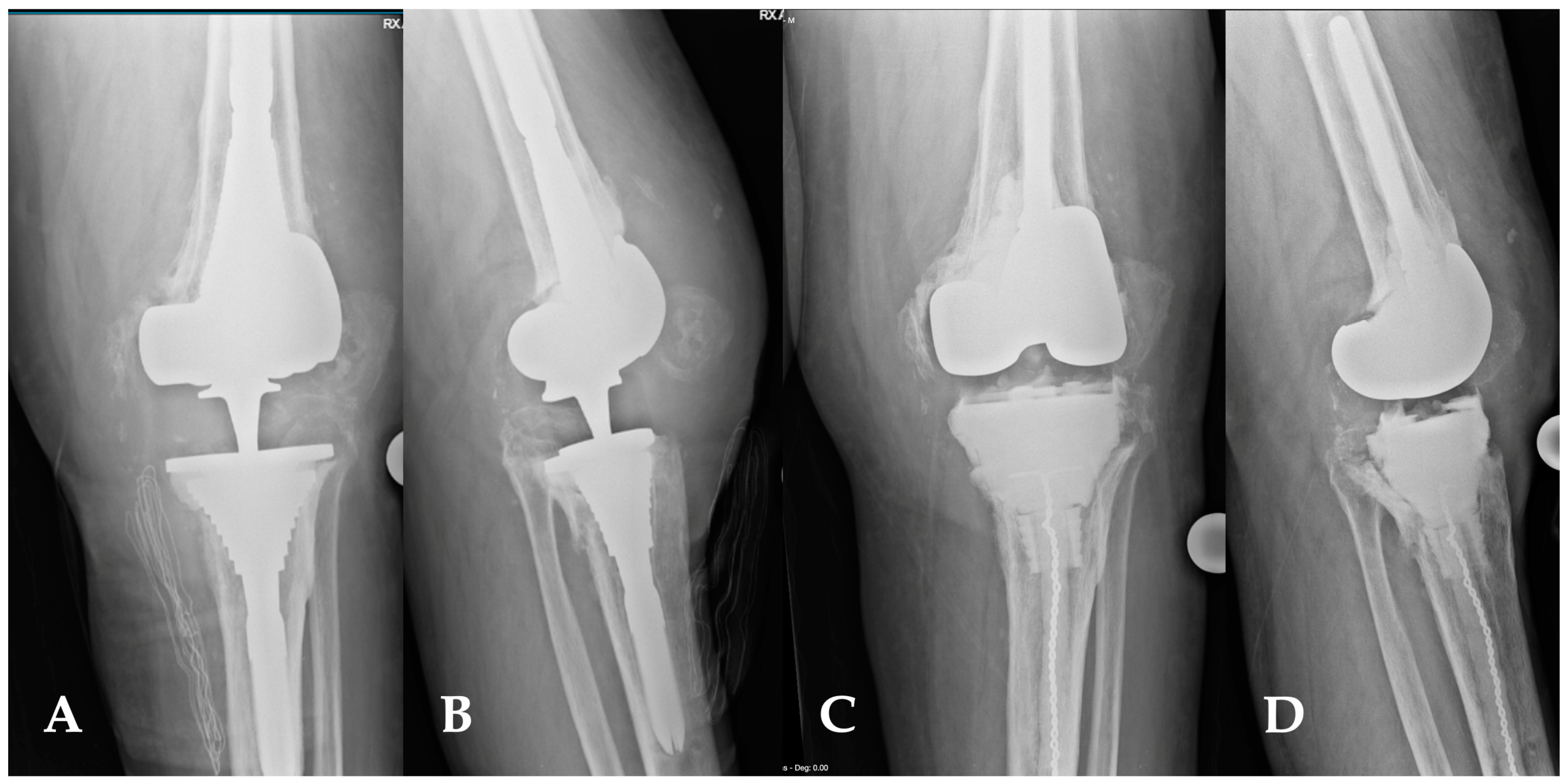
Figure 9.
(A,D) This image depicts the comparison between preoperative and postoperative X-rays of a patient affected by periprosthetic joint infections of the knee (A,B). The patient’s characteristics met the inclusion criteria for the indication of a balanced articulating spacer. The spacer was constructed using cones and both asymmetrical and symmetrical augments, particularly to address the severe bone loss of the tibia (C,D).
Figure 9.
(A,D) This image depicts the comparison between preoperative and postoperative X-rays of a patient affected by periprosthetic joint infections of the knee (A,B). The patient’s characteristics met the inclusion criteria for the indication of a balanced articulating spacer. The spacer was constructed using cones and both asymmetrical and symmetrical augments, particularly to address the severe bone loss of the tibia (C,D).
[ad_2]